THE STRENGTH OF ACIDS AND ALKALIS
- An acid produces hydrogen ions in aqueous solution. The acidity of a solution is a measure of the concentration of the hydrogen ions in the solution.
- A base produces hydroxide ions in aqueous solution. The alkalinity of a solution is a measure of the concentration of the hydroxide ions in the solution.
- In 1909, Soren Sorensen proposed the pH scale for measuring acidity or alkalinity of an aqueous solution.
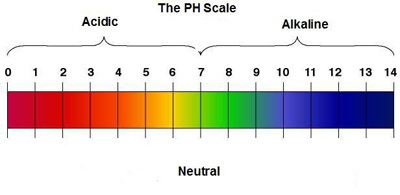
The pH Scale
- The pH scale normally has a range of values from 0 to 14 to indicate how acidic or alkaline an aqueous solution is.
- The pH value measures the concentration of hydrogen ions or hydroxide ions.
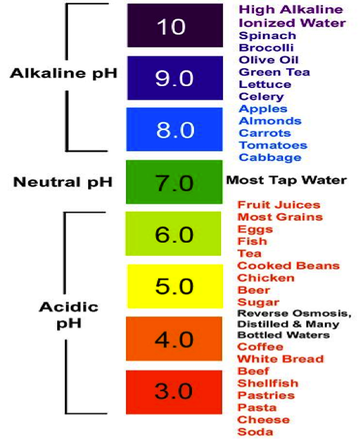
- The pH values of some common solutions used in daily life are shown below.
- The pH value of an aqueous solution can be measured by using
- pH meter
- acid-base indicators
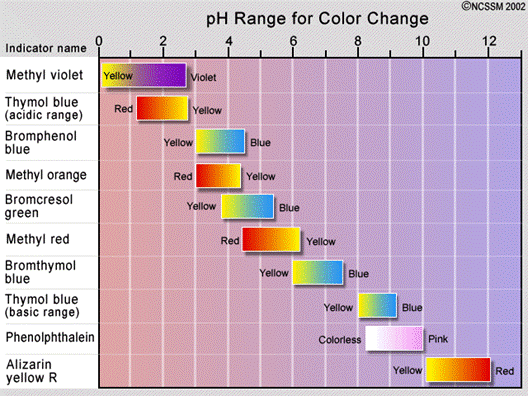
- Other indicator that commonly used are
- methyl orange
- phenolphthalein
- methyl red
- The chart below shows the pH range for some indicators used.
Strong and Weak Acids
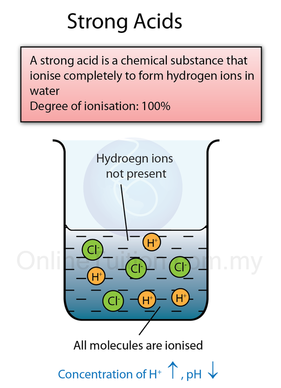
- A strong acid is an acid which ionises or dissociates completely in water to produce a high concentration of hydrogen ions.
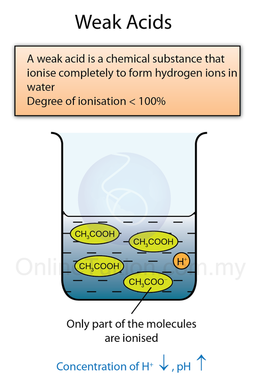
- A weak acid is an acid which ionises partially in water to produce a low concentration of hydrogen ions.
Strong and Weak Alkalis
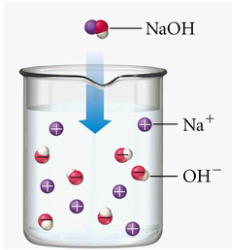
- A strong alkali is an alkali which is fully ionised in water to produce a high concentration of hydroxide ions.
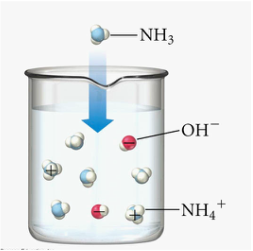
- A weak alkali is an alkali which ionises partially in water to produce a low concentration of hydroxide ions.
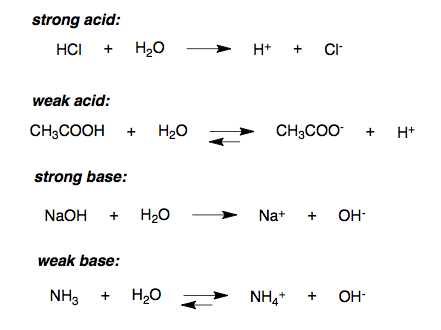
- The figure below shows the chemical equation for dissociation/ ionisation of strong acid, weak acid, strong base and weak base in water respectively.
RELATED RESOURCES
You may try click on the following related resources to get more information.
You may try click on the following related resources to get more information.
- More Ideas about Acids and Bases by Chem4kids