In the chemistry classroom, the students must able to use and comprehend three levels of representation, which are macroscopic, submicroscopic and symbolic representations (Johnstone, 1993). These three levels of representation are related to each other in the explanation of chemistry concepts. Macroscopic refers to what can be observed using the human senses of sight, smell, touch, and hearing. Submicroscopic refers to what scientists believe is actually taking place at the particulate level (atoms, ions, and molecules) in a chemical reaction. Human naked eyes cannot observe the breaking or formation of chemical bonds. Human can only observe the macroscopic evidence that chemical and physical changes are occurring at the submicroscopic level. It is these changes that occur at the particulate level that students have difficulty comprehending and relating to their macroscopic observations. Symbolic representation refers to the chemical symbols found on the periodic table and other symbols used in writing chemical formulae and equations (Muhammad, 2002). Since students do not fully understand chemical occurrences at the submicroscopic level, the symbols and formulas in chemical equations lack sufficient meaning. Hence, simulation plays an essential role in an attempt to bring abstract chemistry topics into a concrete and understandable form.
|
You can try out this simulations to measure the pH of some common solutions in our daily life.
You may view or download this simulation online. |
|
|
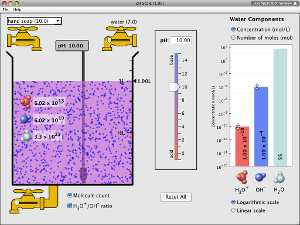
A similar simulation for the pH measurement. You can simply change the concentration of solution by adding in the water in the tank provided.
You may view or download this simulation online. |
|
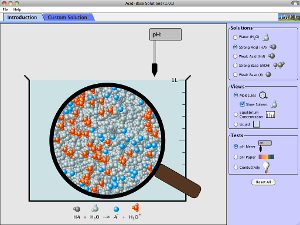
You may try this simulation to - relate concentration of hydrogen ions with pH value - relate concentration of hydroxide ions with pH value - relate strong or weak acid with degree of dissociation - relate strong or weak alkali with degree of dissociation - conceptualise qualitatively strong and weak acids - conceptualise qualitatively strong and weak alkalis You may view or download this simulation online. |